|
11/28/2023 0 Comments C2 molecular orbital diagramIn general chemistry courses, students learn that covalent bonds can come as either single, double or triple bonds, which are identifies by their bond order. To explain the observed paramagnetic properties of molecular oxygen with Molecular Orbital theory.To describe the connection between bond order, bond length and bond energy in diatomic molecules.Magnetic properties: Since each 2p x and 2p y MO contains unpaired electron, therefore B 2 molecule is paramagnetic.\) The two boron atom is B2 molecules are linked by one covalent bond. 6 in all, have to be accommodated in various molecular orbitals in the increasing order of their energies. In the formation of B 2 molecule, three valence electrons of each boron atom i.e. 
A number of valence electrons of each boron atom = 3. It is diamagnetic due to the absence of any unpaired electron.ī 2 molecule: The electronic configuration of B atom (Z = 5) isī 2 molecule is formed by the overlap of atomic orbitals of both boron atoms. Magnetic property: Since bond order is zero, Be 2 molecule does not exist. The molecular orbital electronic configuration, 4 in all, have to be accommodated in various molecular orbitals in the increasing order of their energies. Thus in the formation of Be 2 molecule, two outer electrons of each Be atom i.e. Number of valence electrons in Be atom = 2 (i) Be2 molecule: The electronic configuration of Be(Z = 4) is:īe 2 molecule is formed by the overlap of atomic orbitals of both beryllium atoms. (iv) Magnetic character: As it has two unpaired electrons one each in orbitals, it is paramagnetic. Since bond order is positive, it is quite stable. (c) ion: It is formed when molecule gains two electrons, (iv) Magnetic character Due to the presence of an unpaired electron in *2p x orbital, it is paramagnetic. Bond order being positive, N 2 is quite stable. This electron will go to either orbital of N 2 each of which is empty. (b) ion : It is formed by the gain of one electron by N 2 molecule. (iv) Magnetic character: Since 2p z orbital has one unpaired electron, therefore it is paramagnetic. (iii) Stability: As the bond order is positive, it is quite stable. This electron will be lost from 2p 7 orbital of nitrogen molecule. However, if the bond order is the whole number, the molecule may or may not be paramagnetic.Ī) ion: It is formed from a molecule by the loss of one electron. (v) If the bond order is fractional, the molecule will definitely be paramagnetic. Bond order of a molecule is equal to the number of covalent bonds between the atoms in the molecule. 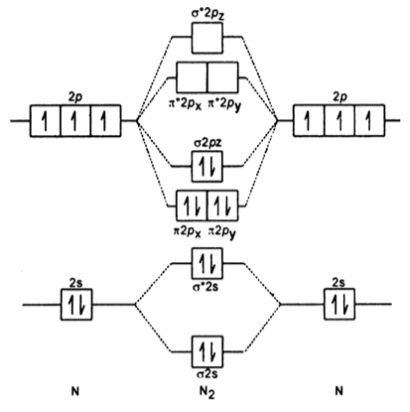
(iv) Knowing the bond order, the number of covalent bonds between the atoms in the molecule can be predicted. greater the bond order, the shorter will be the bond length. (iii) Bond length of the molecule is inversely proportional to the bond order of the molecule i.e. greater the bond order, greater is the bond dissociation energy. (ii) The dissociation energy of the molecule is directly proportional to the bond order of the molecule i.e. (i) If the value of the bond order is positive, it indicates a stable molecule and if the value of the bond order is negative or zero, it means that the molecule is unstable and is not formed. Bond order may be defined as half the difference between number of electrons in bonding molecular orbitals and the number of electrons in antibonding molecular orbitals, i.e.,
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
